Volume: 19 Issue: 1 January 2021
FULL TEXT
Objectives: There is an increased risk of vascular complications in kidney transplant for allografts with multiple renal arteries versus a single renal artery. We compared the clinical outcomes of living donor kidney transplant recipients who received allografts with a single renal artery versus multiple renal arteries.
Materials and Methods: This retrospective analysis included all living-related donor kidney transplants that were performed by a single skilled urologist. All donor nephrectomies were performed by open method. The left kidney was preferred over the right for donor nephrectomy, except in cases of vascular problems or other contraindications, for which the right kidney was preferred. In most of the cases, kidneys were placed in the right iliac fossa for transplant by an extraperitoneal approach.
Results: Of 97 living donor kidney transplants, 82 had a single renal artery (group 1) and 15 had multiple renal arteries (group 2). Patients ranged in age from 18 to 76 years old. Recipient ages (33.00 vs 29.46 years) and baseline serum creatinine values (8.61 vs 8.82 mg/dL) were comparable in groups 1 and 2 (P > .05). However, mean operative time and total ischemia time were significantly higher in the multiple renal artery group (221 and 53.45 minutes, respectively) compared with the single renal artery group (202 and 77.6 minutes, respectively). Graft survival at 1 year was 95.12% in the single renal artery group and 93.33% in the multiple renal artery group. Patient survival at 1 year was 96.34% in the single renal artery group and 93.33% in the multiple renal artery group.
Conclusions: The safety of kidney transplants of allografts with multiple renal arteries is equal to the safety of transplants of allografts with a single renal artery in terms of vascular complications and acute tubular necrosis, as well as patient and graft survival.
Key words : Graft survival, Vascular complications
Introduction
Living donor kidney transplant has been practiced for the past 5 decades in India.1 End-stage renal disease, regardless of the etiology, is the primary indication for kidney transplant. Among all the treatment interventions, kidney transplant is favored because of less morbidity and a better quality of life.2 Kidney transplant techniques have evolved through the years, which has led to drastic improvements in graft and patient survival rates.3,4
Before transplant, each donor is assessed for renal vascular abnormalities with computerized tomography or magnetic resonance imaging.5 The most frequent vascular abnormality observed in donors is the presence of multiple renal arteries (MRAs), the prevalence of which is 18% to 30% as reported in the literature.6,7 The presence of MRAs varies according to ethnicity and fluctuates from 4% in Malaysians to 59.5% in Indians, which differs with the type of renal artery, and is lowest in people from eastern and southern Asia (from 4% to 18.4%).8,9 The type of visualization technique and stage of morphological growth also determine the frequency of MRAs.10
Studies have demonstrated that kidney transplants with a single renal artery (SRA) are ideal because of fewer complications. Kidneys with MRAs are reported to have a higher potential to cause impaired clinical outcomes after kidney transplant.11 Vascular complications such as arterial thrombosis and renal artery stenosis12 due to MRAs are serious events that affect patient and graft survival in renal transplant. Prolonged ischemia time and delayed graft function (DGF) are other complications that may occur in transplants of kidneys that have MRAs.13,14 Despite this fact, some studies propose that kidney transplants of allografts with MRAs are as safe as those with SRA.6,11
Efforts have been made to condense the occurrence of MRA-associated vascular complications.15 Revascularization is one such technique that can be performed simultaneously or sequentially. Anatomic abnormalities in renal grafts do pose difficulty during surgery; therefore, implantation of kidneys with MRAs requires an experienced surgeon to perform vascular reconstruction with speed and precision, to improve total ischemia time without compromising the vascular anastomosis. Although there may be an increase in ischemia time in some cases with MRAs, damage to the graft may be avoided if the anastomosis is performed within reasonable speed and with proper cooling of the graft.16
In the present study, living donor kidney transplant with MRAs versus SRA was reviewed retrospectively, and the short- and long-term outcomes were compared.
Materials and Methods
Study design
We conducted a retrospective study in patients who received a living donor
kidney between January
2012 and August 2018 in a regional transplant
center. Deceased donor kidney transplants and
ABO-incompatible cases were excluded from the study. The study protocol was
approved by the ethics committee of our hospital (approval No. MGM-ECRHS/2018/59
dated 5.10.2018) and conducted
in accordance with principles of the Declaration of Helsinki and International
Council for Harmonization-Good Clinical Practice. Detailed clinical history was
recorded, and physical examination was performed on the recipients. Computed
tomography angiography was performed for evaluation of donor renal
vascular abnormalities. Living donors were evaluated for human leukocyte antigen
typing, and tissue crossmatch was checked between donors and recipients before
transplant. All donors and
recipients were checked for ABO blood group compatibility.
All kidney transplants were performed by a single trained and skilled urologist (ADM), with the aid of ×2.5 magnifying loupes. All donor nephrectomies were performed with the open method. The left kidney was preferred over the right for donor nephrectomy, except in cases of vascular problems or any other contraindications for which the right kidney was preferred. In all cases, kidneys were placed in the right iliac fossa for implantation by an extraperitoneal approach, except in 3 cases of second transplant in which the graft was placed in the left iliac fossa.
The renal vein was anastomosed to the external iliac vein in an end-to-side manner. In cases of SRA, the renal artery was most often anastomosed to the internal iliac artery in an end-to-end manner. In MRA cases, the major renal artery was anastomosed to the internal iliac artery in an end-to-end manner, and the other renal arteries were anastomosed to the external iliac artery by the end-to-side technique. Fine polypropylene sutures (Prolene 6-0) were used for vascular anastomoses. For smaller caliber arteries, 7-0 Prolene sutures were used for anastomosis. A standard Lich-Gregoir ureteroneocystostomy was performed in all cases with 6-0 PDS polydioxanone absorbable sutures. A double J stent was not routinely placed during ureteroneocystostomy. A double J stent was only inserted in select patients with neurogenic bladders, ureteral abnormalities, small stones in the donor kidneys, and reduced or no immediate urine output after the vascular anastomosis. The drain was removed if it was not draining for 24 hours, and the urethral catheter was removed usually on the fifth postoperative day. Doppler ultrasonography was routinely performed in all patients at the seventh day posttransplant.
All recipients were given prophylactic broad-spectrum parenteral antibiotics in the operating room during transplant along with optimal hydration. Recipients were given 500 mg and 125 mg methylprednisolone intraoperatively and in the evening on the day of transplant. Furthermore, these patients received 20 mg prednisolone for the first month, 15 mg for second month, and 10 mg after the third month. Tacrolimus was given at a dose of 0.12 mg/kg/day, and we monitored tacrolimus serum levels to avoid toxicity. Mycophenolate mofetil was started at a dose of 2 g for 3 months and was given at a dose of 1.5 g thereafter. We used cotrimoxazole and valganciclovir for prophylaxis.
Outcomes
The postoperative outcomes that we studied included recipient complications and
baseline and postoperative serum creatinine, total ischemia time, mean operating
time, and short-term and long-term graft and patient survival.
Statistical analyses
We used Kaplan-Meier analysis to compute the survival function. Qualitative and
quantitative data are presented as number of patients (with % of total) and mean
values (with SD). We compiled the data in a master chart (a Microsoft Excel
spreadsheet) and used SSPS software (version 25; IBM) to analyze the data.
Qualitative data are presented as frequencies and percentages. Mean values (with SD) were calculated for quantitative variables. We applied the chi-square test to check significant associations between study groups and outcome. We applied the unpaired t test to check significant differences between the 2 study groups. The significance level was P < .05.
Results
Of the 97 living donor kidney transplants, 82 had SRA (group 1) and 15 had MRA (group 2). Within group 2, only 1 patient had 3 renal arteries and 14 patients had double renal arteries. The patients ranged in age from 18 to 76 years. Both groups were compared with respect to age, baseline creatinine level, mean operative time, and ischemia time. Recipient age (33.00 vs 29.46 years) and baseline serum creatinine (8.61 vs 8.82 mg/dL) were comparable in groups 1 and 2 (P > .05). However, the mean operative time and the total ischemia time were significantly higher in the MRA group (221.00 and 53.45 minutes, respectively) compared with the SRA group (202.00 and 77.60 minutes, respectively). Table 1 shows the preoperative and intraoperative recipient parameters. One patient with SRA (group 1) required immediate intraoperative revision of arterial anastomosis to mitigate a technical problem. Of the 3 patients requiring a second transplant, 1 patient with MRA (2 arteries) required the graft to be placed in the left iliac fossa. Hence, it was a technical challenge to perform 3 vascular anastomoses (2 arterial and 1 venous) in the left iliac fossa of this patient.
In early postoperative outcomes, DGF, which is defined as need for dialysis within 1 week of transplant, was 4.87% in group 1 and 13.33% in group 2 (P = .21). The rates of renal artery stenosis diagnosed by color Doppler and both angiographies were 3.65% and 6.66% in groups 1 and 2, respectively (P = .59). Biopsy-proven cases of acute tubular necrosis were higher in group 2 (13.33% vs 9.75%). Likewise, acute rejection in group 2 was 6.66% and in group 1 was 2.43% (Table 2).
Long-term outcomes of transplant in recipients were compared in terms of the mean serum creatinine at 6 months and at 1 year of follow-up, which were comparable in both groups (P = .055 and .133). Graft survival at 1 year was 95.12% in the SRA group and 93.33% in the MRA group. Patient survival at 1 year was 96.34% in the SRA group and 93.33% in the MRA group (Figure 1). Thus, long-term outcomes were similar in both groups (Table 3).
Discussion
Despite medical advancements in transplant techniques, grafts with MRAs are contraindicated because of vascular and urologic complications. In this retrospective study, we compared the outcomes of kidney transplant of allografts with MRAs versus those with SRA.
Although the literature has reported complications associated with MRAs and discourages their use,14 other investigations have suggested successful kidney transplants with MRAs.6,17 Likewise, the present study also showed comparable results between the allografts with SRA and MRAs in terms of baseline creatinine, mean operative time, and ischemia time. Survival rates of patient and grafts at 1 year were comparable in both groups. Long-term outcomes and safety indexes were analogous in both groups.
At the present center, only living-related kidney transplants are performed, and the majority of donors are first-degree relatives. As a rule, unrelated transplants are not performed at this center. Hence, for our study, the pool of available donors was limited; therefore, we accepted donors with all types of vascular abnormalities and comorbid conditions, within reasonable limits.
Complex vascular anatomy poses a challenge to the surgical skills of the operating team. The incidence of MRAs is considered a common anatomic variation of the kidneys, which deviates with each study. In the present study the incidence of MRAs was found to be 15.46%, which is lower than the value of 30% reported by Aydin and colleagues (2004)17 and higher than the value of 8.3% reported by Kamali and colleagues (2012).18
A study by Usta and colleagues (2015)19 was conducted to evaluate the complications of MRAs and long-term kidney function in patients of similar age as enrolled in the present study; however, the mean follow-up time in that study was 22.5 months in contrast to 12 months in the present study. Usta and colleagues concluded that the differences with regard to occurrence of vascular and urologic complications and DGF were insignificant in grafts with MRAs compared with grafts with SRA.19
In a study by Saidi and colleagues,11 the data of 350 consecutive kidney transplants comprising 319 renal allografts with SRA and 31 allografts with MRAs showed shorter operative time in SRA grafts compared with MRA grafts (173 vs 259 minutes), which is analogous to the present study. Saidi and colleagues concluded that there has been a significant increased use of allografts with MRAs in recent years, especially in grafts procured by laparoscopy, and this increase may be the result of the proven feasibility and safety associated with MRAs.11A large cohort study20 of 2100 patients in a single tertiary center during a period of 33 years reported the prevalence, demographics, and surgical techniques for transplants of kidneys with MRAs; that study20 showed grafts with MRAs were associated with a prolonged ischemia time (P = .001) and acute tubular necrosis, similar to the present study. Regardless of these outcomes, rates of survival of patients or grafts were not adversely affected.20
Multiple renal arteries have been reported to be associated with arterial stenosis and thrombosis. Also, inadvertent ligation of polar arteries may cause urologic complications such as ureteral necrosis and calyceal or ureteral fistulas.21 Extracorporeal microsurgical bench reconstruction of MRAs has led to improved management of renal transplant with MRAs.22,23 However, this procedure was not performed in any of the cases in the present investigation. In cases of multiple anastomoses, revascularization can be performed either simultaneously with the entire arterial engraftment or sequentially after the engraftment. For the latter technique, revascularization is performed first in the main renal artery. The vascular clamps are released and the kidney is partially revascularized until the other artery is anastomosed, while the renal artery perfusion is being sustained.6 However, at this center, the clamps were released after all the arterial anastomoses were completed in the MRA group.
Kadotani and colleagues (2005)16 and Moreno-Alarcón and colleagues (2013)24 reported a higher but nonsignificant incidence of DGF in MRA allografts. This observation is similar to the observation in the present study in which a higher but nonsignificant incidence of DGF was reported (3.65% vs 6.66%; P = .21).
In this study, postoperative outcomes, such as acute rejection, creatinine levels, surgical complications, and graft survival, between the 2 groups were comparable. Many earlier studies have also shown similar results.25-27 Ashraf and colleagues (2013)6 concluded that kidney transplants of allografts with MRAs are as safe and successful as transplants with SRA with regard to vascular and urologic complications and survival rates of patients and grafts. The rates for 1-year and 5-year graft survival were 93% and 78%, respectively, in MRA allografts and 95% and 73%, respectively, in SRA allografts (P = .89).17 In another large cohort study of 201 patients with a mean follow-up period of 10 years, Ghazanfar and colleagues28 concluded that allografts with MRAs were safe and showed survival rates for grafts and patients that are in agreement with the present study; however, the patients in that study from 2010 experienced vascular complications of 8.9% and a higher incidence of acute tubular necrosis and posttransplant hypertension (4%),28 which were not reported in this present study. Ali-El-Dein and colleagues (2003)29 reported higher mean serum creatinine values at 1-year follow-up in grafts with MRAs, and this was attributed to longer ischemia time. However, in the present study, serum creatinine at 1 year was comparable in both groups. In contrast, Osman and colleagues (2003)30 reported unfavorable results with MRAs; they concluded that grafts with MRAs were associated with an increased rate of thrombotic and stenotic complications.
A meta-analysis of 23 studies, comprising 18 289 patients who received MRA grafts, showed significantly higher complication rates (13.8% vs 11.0%; odds ratio 1.393, P < .0001) and significantly more DGF (10.3% vs 8.2%). Despite higher complications with MRA grafts, the long-term survival rates (for grafts and patients) with MRA allografts were similar to the survival rates associated with SRA allografts.14
Previously published studies reported frequent incidence of lymphoceles (12.5%) with MRAs,31 whereas there was no such MRA-associated rate of lymphocele incidence in the present study. An Indian study reported patient survival rates similar to ours, and none of the patients developed vascular or urologic complications after receiving an allograft with MRAs, which confirmed transplant outcomes with rates of success similar to SRA.32 This study and other investigations from the literature showed no association between multiplicities of renal arteries and the risk of vascular complications.
The present study has limitations such as small sample size and single center. Also, because the type of population is an important factor for variation in outcomes of kidney transplant, it is not appropriate to extrapolate these results to a larger population of diverse ethnicity.
Conclusions
Kidney transplant of allografts with MRAs is technically challenging; however, the use of allografts with MRAs was found to be as safe as with SRA with respect to vascular complications and patient and graft survival.
References:
- Abraham G, John GT, Sunil S, Fernando EM, Reddy YN. Evolution of renal transplantation in India over the last four decades. NDT Plus. 2009;3(2):203-207. doi:10.1093/ndtplus/sfp178
CrossRef - Emiroglu R, Koseoglu F, Karakayali H, Bilgin N, Haberal M. Multiple-artery anastomosis in kidney transplantation. Transplant Proc. 2000;32(3):617-619. doi:10.1016/s0041-1345(00)00919-2
CrossRef - PubMed - Bessede T, Droupy S, Hammoudi Y, et al. Surgical prevention and management of vascular complications of kidney transplantation. Transpl Int. 2012;25(9):994-1001. doi:10.1111/j.1432-2277.2012.01533.x
CrossRef - PubMed - Ng ZQ, Lim W, He B. Outcomes of kidney transplantation by using the technique of renal artery anastomosis first. Cureus. 2018;10(8):e3223. doi:10.7759/cureus.3223
CrossRef - Ikidag MA, Uysal E. Evaluation of vascular structures of living donor kidneys by multislice computed tomography angiography before transplant surgery: Is arterial phase sufficient for determination of both arteries and veins? J Belg Soc Radiol. 2019;103(1):23. doi:10.5334/jbsr.1719
CrossRef - PubMed - Ashraf HS, Hussain I, Siddiqui AA, Ibrahim MN, Khan MU. The outcome of living related kidney transplantation with multiple renal arteries. Saudi J Kidney Dis Transpl. 2013;24(3):615-619. doi:10.4103/1319-2442.111087
CrossRef - PubMed - Zhang Q, Ji Y, He T, Wang J. Ultrasound-guided percutaneous renal biopsy-induced accessory renal artery bleeding in an amyloidosis patient. Diagn Pathol. 2012;7:176. doi:10.1186/1746-1596-7-176
CrossRef - PubMed - Palmieri BJ, Petroianu A, Silva LC, Andrade LM, Alberti LR. Study of arterial pattern of 200 renal pedicle through angiotomography. Rev Col Bras Cir. 2011;38(2):116-121. doi:10.1590/s0100-69912011000200009
CrossRef - PubMed - Gulas E, Wysiadecki G, Szymanski J, et al. Morphological and clinical aspects of the occurrence of accessory (multiple) renal arteries. Arch Med Sci. 2018;14(2):442-453. doi:10.5114/aoms.2015.55203
CrossRef - PubMed - Gulas E, Wysiadecki G, Cecot T, et al. Accessory (multiple) renal arteries -Differences in frequency according to population, visualizing techniques and stage of morphological development. Vascular. 2016;24(5):531-537. doi:10.1177/1708538116631223
CrossRef - PubMed - Saidi R, Kawai T, Kennealey P, et al. Living donor kidney transplantation with multiple arteries: recent increase in modern era of laparoscopic donor nephrectomy. Arch Surg. 2009;144(5):472-475. doi:10.1001/archsurg.2009.49
CrossRef - PubMed - Srivastava A, Kumar J, Sharma S, Abhishek, Ansari MS, Kapoor R. Vascular complication in live related renal transplant: An experience of 1945 cases. Indian J Urol. 2013;29(1):42-47. doi:10.4103/0970-1591.109983
CrossRef - PubMed - Mandal AK, Cohen C, Montgomery RA, Kavoussi LR, Ratner LE. Should the indications for laparascopic live donor nephrectomy of the right kidney be the same as for the open procedure? Anomalous left renal vasculature is not a contraindication to laparoscopic left donor nephrectomy. Transplantation. 2001;71(5):660-664. doi:10.1097/00007890-200103150-00015
CrossRef - PubMed - Zorgdrager M, Krikke C, Hofker SH, Leuvenink HG, Pol RA. Multiple renal arteries in kidney transplantation: a systematic review and meta-analysis. Ann Transplant. 2016;21:469-478. doi:10.12659/aot.898748
CrossRef - PubMed - Hashimoto Y, Nagano S, Ohsima S, et al. Surgical complications in kidney transplantation: experience from 1200 transplants performed over 20 years at six hospitals in central Japan. Transplant Proc. 1996;28(3):1465-1467.
PubMed - Kadotani Y, Okamoto M, Akioka K, et al. Renovascular reconstruction of grafts with renal artery variations in living kidney transplantation. Transplant Proc. 2005;37(2):1049-1051. doi:10.1016/j.transproceed.2005.01.033
CrossRef - PubMed - Aydin C, Berber I, Altaca G, Yigit B, Titiz I. The outcome of kidney transplants with multiple renal arteries. BMC Surg. 2004;4:4. doi:10.1186/1471-2482-4-4
CrossRef - PubMed - Kamali K, Abbasi MA, Ani A, Zargar MA, Shahrokh H. Renal transplantation in allografts with multiple versus single renal arteries. Saudi J Kidney Dis Transpl. 2012;23(2):246-250.
PubMed - Usta S, Soyer V, Sarici B, et al. Multiple renal arteries in patients with kidney transplantation: initial experiences of the new kidney transplant center. First Med J. 2015;20:25-28.
- Soliman SA, Shokeir AA, Kamal AI, et al. Long-term outcome of grafts with multiple arteries in live-donor renal allotransplantation: Analysis of 2100 consecutive patients. Arab J Urol. 2011;9(3):171-177. doi:10.1016/j.aju.2011.07.006
CrossRef - PubMed - El Atat R, Derouiche A, Guellouz S, Gargah T, Lakhoua R, Chebil M. Surgical complications in pediatric and adolescent renal transplantation. Saudi J Kidney Dis Transpl. 2010;21(2):251-257.
PubMed - Basaran O, Moray G, Emiroglu R, Alevli F, Haberal M. Graft and patient outcomes among recipients of renal grafts with multiple arteries. Transplant Proc. 2004;36(1):102-104. doi:10.1016/j.transproceed.2003.11.012
CrossRef - PubMed - Makiyama K, Tanabe K, Ishida H, et al. Successful renovascular reconstruction for renal allografts with multiple renal arteries. Transplantation. 2003;75(6):828-832. doi:10.1097/01.TP.0000054461.57565.18
CrossRef - PubMed - Moreno-Alarcón C, Server-Pastor G, Lopez-Gonzalez PA, et al. Must we still be worried about multiple arteries in kidney transplantation? Nephrourol Mon. 2013;5(1):692-696. doi:10.5812/numonthly.4928
CrossRef - PubMed - Wang L, He F, Shi M, Lu Y, Yang Y. [Treatment of multiple arteries in renal transplantation from living related donors]. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2008;22(5):582-585.
- Bakirtas H, Guvence N, Eroglu M, et al. Surgical approach to cases with multiple renal arteries in renal transplantation. Urol Int. 2006;76(2):169-172. doi:10.1159/000090883
CrossRef - PubMed - Gawish AE, Donia F, Samhan M, Halim MA, Al-Mousawi M. Outcome of renal allografts with multiple arteries. Transplant Proc. 2007;39(4):1116-1117. doi:10.1016/j.transproceed.2007.03.067
CrossRef - PubMed - Ghazanfar A, Tavakoli A, Zaki MR, et al. The outcomes of living donor renal transplants with multiple renal arteries: a large cohort study with a mean follow-up period of 10 years. Transplant Proc. 2010;42(5):1654-1658. doi:10.1016/j.transproceed.2009.12.067
CrossRef - PubMed - Ali-El-Dein B, Osman Y, Shokeir AA, Shehab El-Dein AB, Sheashaa H, Ghoneim MA. Multiple arteries in live donor renal transplantation: surgical aspects and outcomes. J Urol. 2003;169(6):2013-2017. doi:10.1097/01.ju.0000067637.83503.3e
CrossRef - PubMed - Osman Y, Shokeir A, Ali-el-Dein B, et al. Vascular complications after live donor renal transplantation: study of risk factors and effects on graft and patient survival. J Urol. 2003;169(3):859-862. doi:10.1097/01.ju.0000050225.74647.5a
CrossRef - PubMed - Mazzucchi E, Souza AA, Nahas WC, Antonopoulos IM, Piovesan AC, Arap S. Surgical complications after renal transplantation in grafts with multiple arteries. Int Braz J Urol. 2005;31(2):125-130. doi:10.1590/s1677-5538200500020000630.
CrossRef - PubMed - Khanna R, Sood R, Madhusoodanan P, et al. Multiple vessel renal transplant: army hospital (research and referral) experience. Indian J Urol.

Volume : 19
Issue : 1
Pages : 38 - 43
DOI : 10.6002/ect.2020.0244
From the Mahatma Gandhi Mission’s Medical College and Hospital, Aurangabad,
India
Acknowledgements: The authors have not received any funding or grants in support
of the presented research or for the preparation of this work and have no
further declarations of potential interest.
Corresponding author: Abhay Mahajan, Department of Urology, MGM Medical College
and Hospital, N-6, CIDCO, Aurangabad 431003, India
Phone: +91 9822321224
E-mail: drabhaymahajan@gmail.com
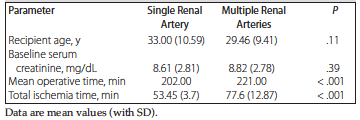
Table 1. Comparison of Preoperative and Intraoperative Recipient Parameters
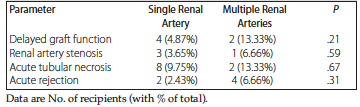
Table 2. Comparison of Early Postoperative Recipient Outcomes
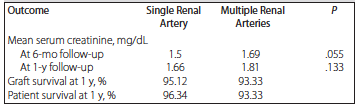
Table 3. Comparison of Long-Term Postoperative Recipient Outcomes
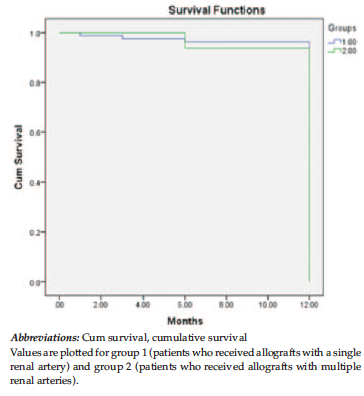
Figure 1. Kaplan-Meier Curve Showing Patient Survival in Both Groups at 1 Year